Electric Vehicle Safety with Advanced EV Battery Cooling
When rapid electrical discharge occurs in an electric vehicle's battery, temperature in a battery cell increases further—this phenomenon is called Battery Thermal Runaway. Learn more about immersion cooling, one solution to help prevent battery thermal runaway, in this video.
For more information, please contact your Lubrizol representative.
Video Transcript
The development of electric vehicles is prolific. And although internal combustion engine innovation continues, the expectation is that electric vehicles will begin to make up a larger portion of the car parc in coming years. One of the most critical safety concerns with electric vehicles is keeping the battery cool during fast charging to avoid overheating and potential vehicle fires.
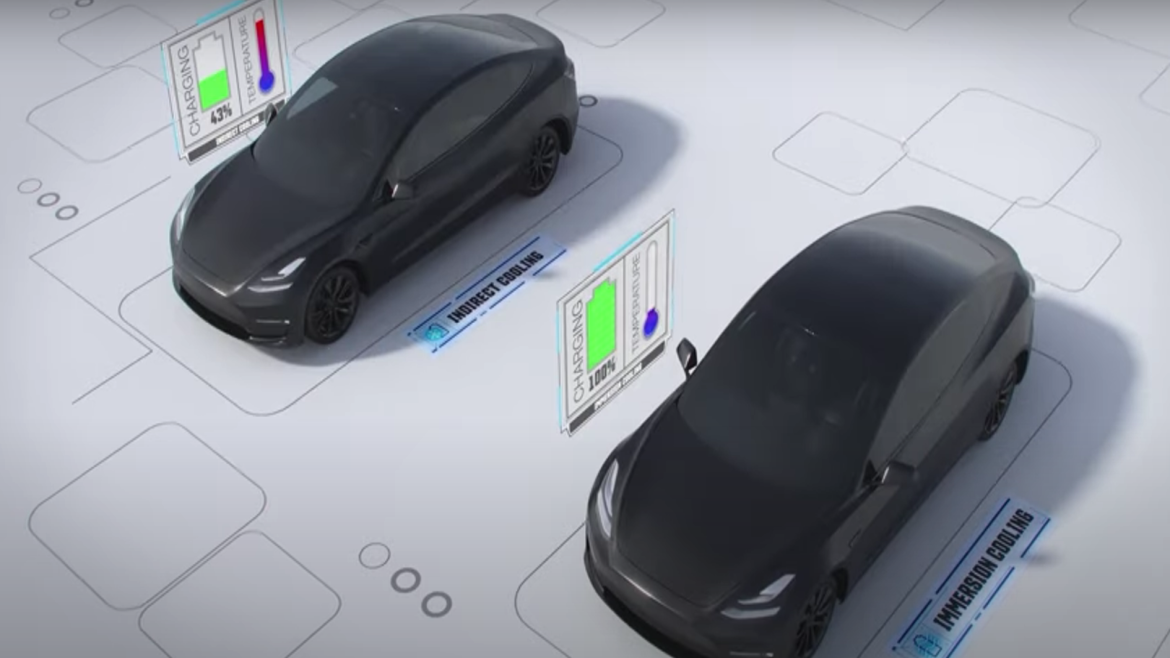
One method of battery cooling currently used is indirect cooling. A cold plate is placed in contact with the battery cells and a water glycol coolant is pumped through the plate, cooling down the batteries. Heat is conducted from the battery through the plate and into the coolant. However, there are some disadvantages to this method. Because the coolant is electrically conductive, it is not in direct contact with the cells. This hinders heat transfer from the cell to the coolant and limits charging speed and vehicle performance. This layer creates a thermal resistance which hinders heat removal.
Let's take a look at a battery cell and why heat can lead to such disastrous consequences. On a basic level, an individual battery cell is comprised of an anode, a cathode, a porous separator, and an electrolyte which fills the cell. The anode and the cathode are the electrodes which contain the active material. The porous separator sits between these electrodes to keep them from coming into electrical contact. If the anode and cathode are ever electrically connected, either by the separator melting or being penetrated by an external object, like a nail, or by a lithium dendrite, rapid discharge of the battery will occur, and the temperature of the cell will rapidly increase, leading to thermal runaway.
Another route to thermal runaway is through the breakdown of the solid electrolyte interface layer, or SEI. The SEI forms at both the interface of both the anode and cathode with the electrolyte. At elevated temperatures, the SEI layer can break down, which leads to the rapid and exothermic decomposition of the electrolyte into gases. This process leads to the release of flammable gases and a rapid increase in battery temperature, a phenomenon called battery thermal runaway.
At a module level, the hot gas spreads throughout the pack, spreading heat to the other cells and causing the SEI layer in other cells to melt, sending them too into thermal runaway. This is called propagation of battery thermal runaway, as one cell entering thermal runaway causes others to fail as well. As more cells undergo thermal runaway, the heat continues to build up within the battery pack. The indirect cooling system cannot keep up and temperatures begin to rise. The structural integrity of the battery pack will be challenged.
Once the battery pack is compromised, air will mix with the flammable gases produced by the failing battery cells. With the correct air to fuel ratio, a simple spark can light the entire battery back on fire in just a few seconds with catastrophic consequences.
One solution to help prevent battery thermal runaway is immersion cooling. The battery is immersed in a fluid, which provides direct contact between the coolant and the battery cells. This method improves the heat removal and can prevent thermal runaway propagating from one cell to the next. Immersion cooling is a safer, more effective way of cooling the battery of an electric vehicle. This is why it is more likely to become more prevalent in upcoming electric vehicle designs.
Learn about Lubrizol's EVOGEN™ thermal management fluids
Read more about EV battery cooling
How Immersion Cooling Can Help Make Electric Vehicle Batteries Safer